|
Ionic complexes undergo rapid ligand-exchange.A preference for more electronegative elements (such as O or F) binding.Tendency to decreasing coordination number across the series.Adoption of coordination numbers greater than 6 (usually 8-9) in compounds. 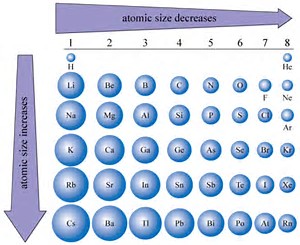 
They can also have an oxidation state of +2 or +4, though some lanthanides are most stable in the +3 oxidation state.Adoption mainly of the +3 oxidation state.Similarity in physical properties throughout the series.These characteristics include the following: Like any other series in the periodic table, such as the Alkali metals or the Halogens, the Lanthanides share many similar characteristics. The term Lanthanides was adopted, originating from the first element of the series, Lanthanum. However, this is can be misleading since the Lanthanide elements have a practically unlimited abundance. These elements were first classified as ‘rare earth’ due to the fact that obtained by reasonably rare minerals. The rest of the elements were later separated from the same mineral. Later, Moseley used an x-ray spectra of the elements to prove that there were fourteen elements between Lanthanum and Hafnium. In 1803, Berzelius and Klaproth secluded the first Cerium compound. 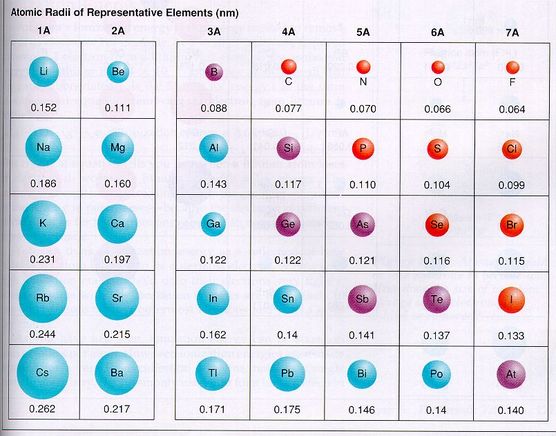
In 1794, Professor Gadolin obtained yttria, an impure form of yttrium oxide, from the mineral. This mineral, now known as Gadolinite, was later separated into the various Lanthanide elements. The Lanthanides were first discovered in 1787 when a unusual black mineral was found in Ytterby, Sweden.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
